What Chemicals Are Popular Among Medicinal Chemists?
Increasing the success rate of high-throughput screening (HTS) and the quality of the resulting hits as well as developability of drug candidates is among the key challenges of small molecule drug discovery programs.
The above goals are associated with so called “compound quality” or “drug-likeness” of the starting small molecules. While the majority of considerations in this context is related to lead-like properties, physicochemical properties, diversity, effective coverage of the chemical space, privileged structures for drugs or structures possessing favourable physical properties or metabolic stability, it was shown in a recent analysis by F.W. Goldberg et al. (2014) that an effective and somewhat overlooked strategy to increase the compound quality is to focus closer on the choice of the building blocks used in the course of drug discovery programs. The nature of the selected building blocks determines not only the speed of the research but also the quality of the resulting drug candidates and their potential in further trials.
Obviously, some reagent classes are more popular than others due to historically synthetic routes adopted by organic and medicinal chemists.
What are medicinal chemists’ favorite reagents?
First of all, it is important to determine the classification principle for building blocks as they can contain several unprotected functional groups. As suggested in the above reference, in the case of several groups, the classification is determined by the one most probably involved in the first synthetic step. For instance, if a building block contains amine and boronic acid groups, it is assigned to the class of boronic acids as the boronate function is likely to be used in the first turn - via suitable and widespread Suzuki-Miyaura coupling or Chan-Lam coupling.
The leadership palm in terms of desirability by medicinal chemists is held by secondary and primary amines, carboxylic acids, boronic acids and boronate esters. Some examples of unusually popular reagents from this group:
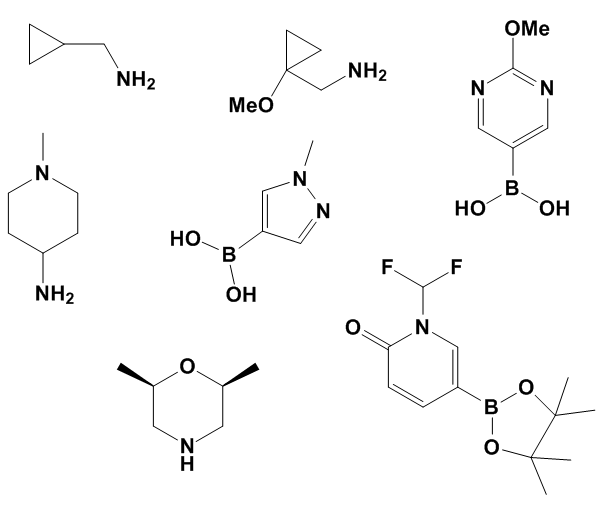
They are followed by aryl halides, alkyl and/or benzyl halides, aldehydes, alcohols and anilines. Somewhat less popular but still widely demanded reagents are ketones, sulfonyl halides and isocyanates, the latter two being useful in Medicinal Chemistry, but given less priority due to their instability during storage.
Building Blocks Supply and Demand: Do They Fit?
Since the industry accepted practice is to purchase building blocks from commercial suppliers, it is interesting to know if currently available offerings are in line with what is demanded by pharmaceutical industry.
One example that might be valuable is a comparison of the distribution by reagent type in a custom synthesis collection created internally by AstraZeneca (customer perspective, blue bars) versus distribution of reagent types in a commercially available offerings (from ACD database, red bars):
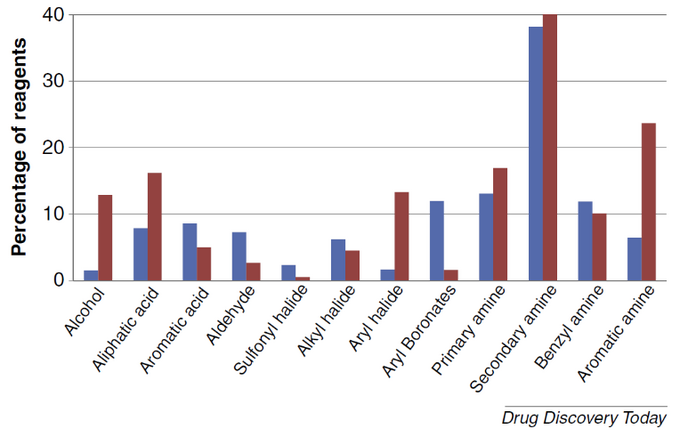
Image from http://www.sciencedirect.com/science/article/pii/S1359644614003894
It is noteworthy that boronates are largely underrepresented in the public catalogs compared to what is expected by medchem teams in the industry.
Topics: Industry Trends
