This Startup Proposes New Lightweight and Informative Molecular Descriptors for Drug Discovery
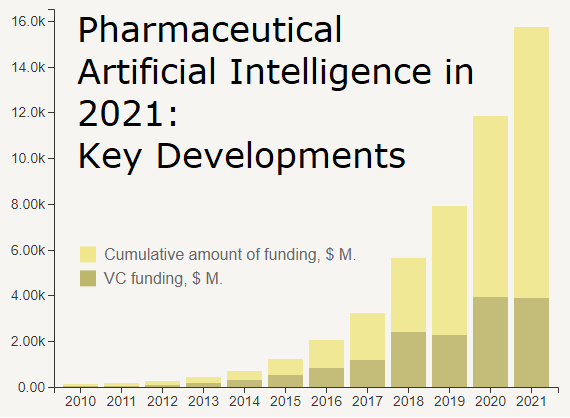
Over the last five or so years, the drug discovery industry has started adopting artificial intelligence (AI) at unprecedented scale, with pretty much every big and small pharma company doing some kind of pilots or more substantial projects having some AI component in it – from machine learning algorithms and deep learning networks to natural language processing models. Technology proved to have such a fundamental impact on performance of drug discovery work, that we now see a wave of young companies – sometimes referred to as "digital biotech" – which have a whole new business model revolving around the platform-based process of innovation. Some companies have "end-to-end" drug design platforms capable of automatically doing not only concept creation and target discovery, but also hit discovery, part of lead optimization work, and even predicting clinical trial outputs and identifying clinically-relevant biomarkers.
Topics: AI & Digital