Lantern Pharma Advances Development of Molecular Diagnostic for Oncology Clinical Trials
Lantern Pharma (NASDAQ: LTRN), a clinical-stage biopharmaceutical company, has reached a milestone in the development of a diagnostic tool for its drug candidate LP-184. This diagnostic, which utilizes quantitative real-time polymerase chain reaction (qRT-PCR) technology, is designed to measure PTGR1 RNA levels in patient tumor samples, thereby assessing their potential sensitivity to LP-184.
Development and Validation
The qRT-PCR based diagnostic aims to quantify PTGR1 RNA to determine tumor sensitivity to LP-184, a precision oncology drug candidate targeting a variety of solid tumors. Lantern Pharma's focus on PTGR1 as a key biomarker has been confirmed, marking a critical step towards creating a companion diagnostic tool that will enhance patient selection for clinical trials. This tool is expected to optimize trial outcomes by selecting patients most likely to benefit from LP-184.
Lantern Pharma is committed to further developing and validating this assay. The company plans to partner with central labs and cancer centers to integrate this diagnostic tool into patient selection and stratification processes for clinical trials. This strategic development aligns with the findings of a 2021 study by Parker et al. in Cancer Medicine, which highlighted the significant positive impact of biomarker use on the success of clinical trials across common cancers.
Biomarker Utilization
Incorporating the PTGR1 biomarker into the development strategy of LP-184 aligns with precision medicine best practices and aims to increase the probability of clinical trial success. PTGR1 levels are higher in certain cancer cells, allowing Lantern Pharma to target cancer indications most likely to respond to LP-184. Research published in October 2023 in Molecular Cancer Therapeutics by Lantern Pharma and collaborators from Fox Chase Cancer Center demonstrated a direct correlation between higher PTGR1 expression and the potency of LP-184, measured in IC50 values.
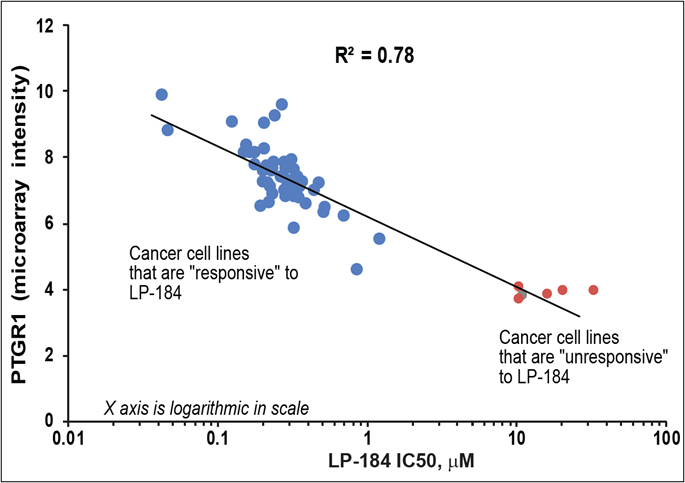
Correlation of PTGR1 expression and cytotoxicity/potency (IC50, µM) of LP-184 (Graphic: Business Wire)
CEO Insights and Future Plans
Panna Sharma, CEO of Lantern Pharma, emphasized the importance of this milestone in advancing precision oncology:
"By working to develop a companion diagnostic for LP-184, we're not just advancing a drug candidate; we're paving the way for more personalized and effective cancer treatments for patients that have the highest likelihood of benefitting from the therapy. The planned use of biomarkers like PTGR1 in our clinical trials exemplifies our commitment to data-driven, patient-centric drug development."
Lantern Pharma plans to implement this assay in forthcoming clinical trials for LP-184. This approach is expected to streamline the development process and increase the likelihood of success. LP-184 is a novel therapeutic in clinical development for malignant gliomas, pancreatic cancer, and atypical teratoid rhabdoid tumors (ATRT). It has also received Orphan Drug Designation and Rare Pediatric Disease Designation from the FDA.
Topics: Tools & Methods