AI-driven Anima Biotech Achieves Milestone with Takeda Illustrating Opportunities in Small Molecule mRNA Drugs Discovery
New Jersey-based Anima Biotech, a leader in the discovery of small molecule mRNA drugs and their mechanisms of action (MOAs) by phenotypic screening with AI-powered image analysis, announced a research milestone in its collaboration with Takeda.
Anima Biotech identifies small molecule drug candidates that selectively modify the translation of mRNA into proteins on ribosomes to modulate the levels of protein produced, to consequently elucidate their MOA in a target space. Research of the company is powered by a custom technology that attaches fluorescent tags to frequently repeating transfer RNA (tRNA) pairs during translation with fluorescent light. One of Anima’s key areas of interest is neurosciences, the subject of an ongoing, up-to-$1.49 billion collaboration with Eli Lilly.
And a partnership with the Japanese pharma giant Takeda also aims to fight neurological diseases. This collaboration was launched in March 2021 to discover and develop a new class of three to six small molecule medicines for genetically-defined neurological diseases, Huntington’s disease in particular, through a partnership that could generate more than $2.4 billion for the seven-year-old biotech. Under the terms of the agreement, Anima Biotech used its mRNA Lightning platform to discover novel mRNA translation modulators against the collaboration targets. And as the milestone in this partnership has been achieved, Yochi Slonim, Anima’s co-founder and& CEO commented that the result demonstrates the advantage of Anima’s approach with small molecule mRNA drugs.
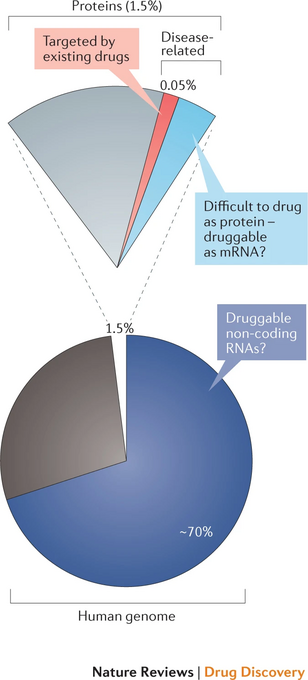
RNA molecules are crucial for delivering cellular information and genetic regulation, but until recently, the drug discovery world has emphasized protein drug targets. However, a shift of focus of the small-molecule therapeutics industry to RNA modulation would expand the landscape of targetable macromolecules by more than an order of magnitude. As shown in the pie chart by Warner, Hajdin et al. in Nature Reviews, only ~1.5% of the genome encodes proteins corresponding to ~20,000 expressed proteins, 10–15% of which are potentially disease-related (~2,000–3,000 proteins encoded by 0.2% of the genome). Currently approved drugs interact therapeutically with <700 of these proteins, using only 0.05% of information encoded by the human genome. Targeting RNAs could expand on the proportion of the human genome that could be therapeutically targeted. Possible RNA targets include mRNAs that encode disease-related proteins that have been characterized as undruggable or difficult to drug as well as non-coding RNAs that influence disease (corresponding to an unknown proportion of the ~70% of the genome that encodes non-coding RNAs).
Up to recently, the lack of knowledge in RNA biology prevented the research on RNA as a drug target, but with such technological advances as sequencing and CRISPR-based techniques, new therapeutic strategies targeting RNA are being explored.
Besides Anima, a number of other companies are pioneering the field. For instance, US-based startup Locanabio that develops RNA targeting gene therapies. The company applies a combination of gene therapy and RNA modification to precisely target disease-causing RNA, using an adeno-associated viral vector to deliver RNA binding proteins into the cells. A startup from Belgium EPICS is developing drugs for unmet medical needs in oncology. Their pipeline includes small molecule drugs that bind to RNA, targeting the epigenetic mechanisms underlying cancer development.